Subcutaneous Implantable Cardioverter Defibrillators Explained


Intro
As the field of cardiac medicine continues to evolve, innovations such as subcutaneous implantable cardioverter defibrillators (S-ICDs) have emerged at the forefront of arrhythmia management. Unlike traditional implantable devices, S-ICDs offer a new approach by being implanted just beneath the skin, eliminating many of the complications associated with transvenous leads. This article will provide a thorough understanding of S-ICDs, charting their design, functionality, clinical usage, and their significance in advancing cardiac care.
Research Overview
Summary of Key Findings
Investigations into S-ICDs have spotlighted several critical insights, namely their effectiveness in detecting life-threatening arrhythmias and delivering appropriate shocks. Clinical studies demonstrate that S-ICDs not only provide reliable protection against sudden cardiac arrest but also come with added benefits such as lower risk of infections and fewer lead-related complications.
Background and Context
The advent of S-ICDs stems from a pressing need to enhance patient outcomes while minimizing risks. Traditional implantable cardioverter defibrillators (ICDs) are often encumbered by complications arising from lead placement within the bloodstream, including lead misplacement, vascular injury, and infection. The evolution to S-ICDs—offering an externalized lead system—marks a significant shift aimed at improving patient safety and quality of life. As arrhythmias evolve, healthcare practitioners must adapt promptly to navigate this shifting landscape in cardiac therapies.
"S-ICDs represent a beacon of innovation in cardioverter therapy, merging technological advancement with clinical prudence, ultimately redefining our approach to arrhythmia management."
Methodology
Experimental Design
The research surrounding S-ICDs has employed various methodologies aimed at understanding their efficacy and safety profile across diverse populations. Most studies utilize a multicenter, prospective cohort design to ensure comprehensive data collection and analysis.
Data Collection Techniques
Data collection often processes through the following channels, ensuring a wealth of information:
- Patient Registries: Ongoing surveillance of patients who have received S-ICDs provides continuous insights into real-world effects.
- Clinical Trials: Multinational trials assess outcomes based on various demographics and clinical presentations.
- Longitudinal Studies: These evaluate the performance and complication rates of S-ICDs over extended periods, tracking both survival rates and quality of life indicators.
This research ultimately establishes a robust foundation for clinicians and patients alike to make informed decisions regarding the deployment of S-ICDs in arrhythmia management.
Intro to Subcutaneous
The world of cardiac care is continually evolving, with advancements thatow so many patients, and one such breakthrough is the introduction of the subcutaneous implantable cardioverter defibrillator (S-ICD). This technology is not just a new gadget; it represents a significant step towards safer and more efficient management of arrhythmias. The S-ICD's design allows for the treatment of patients with a risk of sudden cardiac arrest while minimizing the risk of complications that are often associated with traditional systems, primarily due to its unique placement and operational methodology.
Definition and Purpose
A subcutaneous implantable cardioverter defibrillator is a device implanted beneath the skin, specifically designed to monitor and treat certain types of irregular heartbeats. Unlike traditional devices that are often placed transvenously, the S-ICD operates without the leads in the heart. This fundamental difference not only reduces the likelihood of infection but also makes the device suitable for a wider variety of patients who may be at risk from complications associated with lead placements.
This innovative technology aims to provide fast detection and effective intervention of life-threatening arrhythmias, delivering shocks when necessary to restore a normal heart rhythm. It’s like having a safety net for your heart, ready to act in an emergency, enhancing the quality of life for patients while also providing peace of mind for their families.
Historical Context
Understanding the history of the S-ICD is pivotal in appreciating its development and relevance today. The traditional defibrillator, which involves lead placement inside the heart, has been around for decades and has saved countless lives. However, these devices come with their own set of risks, such as infections and lead dislodgements, which can complicate the patient's experience.
The incremental advancements in medical technology and research highlighted the need for an alternative that could mitigate these risks. As a result, in the early 2000s, the concept for a subcutaneous device began to take shape. After numerous studies and clinical trials, the first S-ICD system was introduced to the market in 2012, heralding a new era of arrhythmia management.
As more data became available, it became clear that this device was not just another option but a genuine improvement in cardiac care. It established new guidelines for patients, particularly those who are intolerant to traditional ICD systems. Over time, acceptance and understanding of the S-ICD have grown among heart specialists, leading to better patient outcomes.
Technological Foundations
Understanding the technological foundations of subcutaneous implantable cardioverter defibrillators (S-ICDs) is key to appreciating their role in contemporary cardiac care. This section illustrates how these devices blend innovation with medical necessity, offering insights on what makes them suitable for certain patient populations compared to traditional ICDs (Implantable Cardioverter Defibrillators).
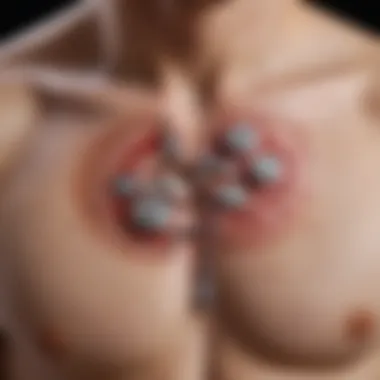
Device Components
The heart of an S-ICD lies in its components. Unlike traditional ICDs that rely on endovascular leads, S-ICDs are designed with innovative parts situated entirely beneath the skin. Key elements include:
- Pulse Generator: This is the powerhouse of the device. It houses the battery and the electronics that detect abnormal heart rhythms.
- Subcutaneous Lead: Instead of being implanted in the heart, this lead sits under the skin and is responsible for delivering shocks when necessary. The lead offers a different approach to rhythm management, making the device less invasive.
- Electrodes: The S-ICD unit has electrodes that monitor the heart's activity. This unique configuration helps in accurate arrhythmia detection by measuring the potential differences caused by electrical activity.
"In essence, the S-ICD's construction speaks to a growing trend in medical device technology: the desire to minimize surgical intrusion while maximizing effective patient outcomes."
Different components come together in a thoughtful manner, creating an integration that supports the core purpose of the S-ICD while addressing the challenges posed by traditional methods. This carefully crafted design reflects ongoing efforts to push the boundaries of cardiac therapy, catering to patients who may be at risk for life-threatening arrhythmias but need a device that ensures safety, comfort, and efficacy.
Operational Mechanics
The operational mechanics of S-ICDs distinguish them further from their traditional counterparts. After implantation, the device's function hinges on its ability to sense arrhythmias effectively. Key operational aspects include:

- Monitoring Algorithms: These algorithms continuously assess the heart's rhythm. In the event of a detected abnormality, they classify the arrhythmia before a shock is delivered. This decision-making process is crucial, as it aids in preventing unnecessary shocks, thus enhancing patient comfort and experience.
- Shock Delivery Mechanism: When a serious arrhythmia is confirmed, the generator sends a signal to the lead to deliver a high-energy shock. This swift action aims to restore normal heart activity, saving patients' lives when they need it the most.
- Communication with External Devices: Many S-ICDs can communicate with healthcare monitoring systems. This feature facilitates remote check-ups, allowing for timely adjustments or interventions without the patient having to make frequent hospital visits.
The elegance of S-ICDs lies not just in their individual parts but in how they work together to provide robust cardiac protection. With advancements in materials, battery life, and algorithms, the technology has demonstrated a commitment to safety and efficacy in pacemaker therapy. Understanding these principles is fundamental for the medical community to harness the true potential of S-ICDs in everyday clinical practice.
Clinical Indications
Understanding the clinical indications for subcutaneous implantable cardioverter defibrillators (S-ICDs) helps illuminate when and why they are a vital piece of cardiac care. As arrhythmias can lead to serious complications, knowing which patients stand to benefit most from these devices is of great importance. The S-ICD is particularly relevant for those who have a history of life-threatening ventricular arrhythmias but are not suitable for traditional transvenous devices. This article segment will detail the qualifications for S-ICD usage within clinical settings, compare it against traditional options, and highlight the benefits that come with its adoption.
Suitability for Patients
Patients who are considered for S-ICD implantation typically fit a specific set of criteria. First and foremost, these individuals generally have an elevated risk for sudden cardiac arrest due to ventricular fibrillation or ventricular tachycardia. Notably, those with existing structural heart disease, such as dilated cardiomyopathy, or prior myocardial infarctions are also potential candidates. However, it is essential to bear in mind that S-ICDs are not for everyone; for instance, patients with a history of significant cardiac structural defects or those who require pacing are better suited for traditional implantable cardioverter defibrillators (ICDs).
Additionally, the patient’s anatomical considerations play a substantial role in determining suitability. S-ICDs are positioned differently, relying on subcutaneous placement, which may not be feasible in patients with extensive tissue abnormalities or after certain surgeries. Hence, a thorough pre-operative assessment is essential in evaluating the individual’s health record and overall condition.
"The S-ICD presents a unique solution for those in need of defibrillation but who also have complex cardiac histories."
Comparison to Traditional ICDs
When juxtaposed with traditional transvenous ICDs, the S-ICD brings some stark differences into play regarding indications and patient profiles. Traditional ICDs typically function by monitoring the heart's rhythms through leads placed inside the heart chambers, making them effective in a variety of scenarios. However, this method may increase the risk of infections, lead displacement, and other complications linked to device invasiveness.
For many patients, especially those who may not require pacing, S-ICDs offer a promising alternative. They provide an important safety net without direct contact with the heart, thus reducing the likelihood of infection or lead-related issues commonly seen with traditional devices. Patients who are not ideal candidates for the transvenous approach—or who are simply looking to avoid the associated risks—may find the S-ICD sets a new standard in care while yielding similar defibrillation effectiveness.
In summary, the suitability of patients for S-ICDs hinges on a mix of clinical history, anatomical considerations, and specific needs for rhythm control. The clear distinctions between S-ICDs and traditional ICDs also reveal that one size does not fit all when it comes to cardiac interventions. Understanding these nuances is crucial for medical practitioners who aim to tailor treatments to the unique dynamics of each patient.
Benefits of Subcutaneous
Subcutaneous implantable cardioverter defibrillators (S-ICDs) have revolutionized cardiac care by offering distinct advantages over traditional devices. These benefits not only enhance the safety and efficacy of arrhythmia management but also provide a more patient-friendly experience. In this section, we will delve into the various significant benefits of S-ICDs, which include a reduced risk of infection, minimized surgical complications, and enhanced patient comfort.
Reduced Risk of Infection
One notable advantage of S-ICDs is the reduced risk of infection. Unlike transvenous defibrillators, which rely heavily on leads placed inside the heart's vessels, S-ICDs are implanted under the skin without direct access to the bloodstream. This features a far more favorable risk profile in terms of infection rates.
For patients, this means a lower chance of facing serious complications such as endocarditis or device-related infections.
"All patients undergoing procedures should be aware of the potential for infections, but S-ICDs typically come with less baggage in this department."
Studies indicate that the risk of lead-related infections significantly diminishes with S-ICDs. This benefit is especially crucial for individuals who may already have compromised immune systems or those who are wary of infection due to previous medical history.
Minimized Surgical Complications
Surgical procedures, regardless of complexity, come with inherent risks. However, S-ICDs tend to present minimized surgical complications. With a simpler implantation technique compared to traditional ICDs, surgeons can avoid navigating complex vascular structures, which can lead to various complications.
The implantation is generally performed under local anesthesia, often resulting in a quicker recovery time. Potential complications, like pneumothorax, which can occur with alternative approaches, are markedly reduced.
Here’s what typically leads to these advantages:
- Less invasive placement allows avoidance of major structures.
- Local anesthesia reduces the risks associated with general anesthesia.
- Lower recovery times encourage faster return to normal activities.
All these factors contribute to a smoother surgical experience, benefiting both surgeons and patients.
Enhanced Patient Comfort
Ultimately, enhanced patient comfort represents a significant perk associated with S-ICDs. These devices are designed with comfort in mind, allowing patients to maintain a more natural range of motion. With no leads in the vascular system and less bulkiness compared to traditional devices, patients report feeling less discomfort post-implantation.
Furthermore, S-ICDs have a unique mechanism that places the device subcutaneously, allowing for better concealment and cosmetic outcomes. Many patients express satisfaction with how unobtrusive the device feels, leading to a positive impact on their psychosocial well-being. This is particularly impactful for younger patients, who may be more conscious about visible medical devices.
In summary, the benefits of subcutaneous ICD devices extend beyond just medical efficacy. A reduced risk of infection, minimized surgical complications, and enhanced comfort create a more positive overall experience for patients, solidifying their role in contemporary arrhythmia management.
Limitations and Challenges
In the evolving field of cardiac care, subcutaneous implantable cardioverter defibrillators (S-ICDs) offer significant advancements in arrhythmia management. However, as with any innovative technology, these devices come with their share of limitations and challenges. Understanding these aspects is crucial for medical professionals, patients, and caregivers as they navigate the complexities of S-ICD deployment and use.
Device Displacement Issues
One palpable concern with S-ICDs is the potential for device displacement. This issue stems from the subcutaneous implantation site, which can experience various movements from normal daily activities. The defibrillator is designed to sit snugly under the skin, but factors like physical exertion, weight fluctuations, or even a bump during sports can lead to shifts in positioning. Such displacements can interfere with the device’s ability to function optimally, potentially rendering it ineffective in emergency situations.
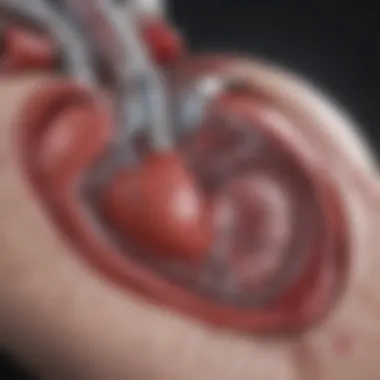

To mitigate this risk, strict adherence to preoperative assessments and careful surgical techniquess are essential. Surgeons often recommend specific post-operative guidelines to reduce the risk of dislodgment, such as avoiding certain physical activities for a period after surgery. Patient education on the signs of displacement can empower individuals to seek timely medical attention, should they notice any irregularities.
Limitations in Arrhythmia Detection
Another notable challenge lies in the limitations of arrhythmia detection associated with S-ICDs. These devices are engineered primarily to detect lethal arrhythmias such as ventricular fibrillation or rapid ventricular tachycardia. However, they may fall short in identifying less critical arrhythmias, which can also pose risks to patients. This can lead to a situation where a patient experiences an episode that the S-ICD does not recognize, creating a false sense of security.
Moreover, the algorithms used for detection are not infallible. Factors like body movement, electromagnetic interference, or changes in chest impedance could affect sensitivity. As a result, there’s an ongoing need for refinement in detection technology to ensure more comprehensive monitoring and intervention. Physicians must remain vigilant, taking a multi-faceted approach to patient assessment and care, encompassing additional monitoring tools or therapies when necessary.
Device Longevity and Battery Life
Finally, the longevity and battery life of S-ICDs are pivotal considerations that can influence their effectiveness. S-ICDs are designed to last several years, with battery life often reported between five to seven years. However, the actual lifespan can be unpredictable, influenced by various factors including device use, how often it delivers shocks, and patient's physiological characteristics.
Once the battery depletes, the need for replacement surgery arises. This procedure can carry its own risks and requires careful planning and scheduling, especially for elderly patients or those with comorbidities. Regular follow-ups are essential to monitor the battery status and ensure that the device remains functional.
Surgical Considerations
When it comes to implanting a subcutaneous implantable cardioverter defibrillator (S-ICD), surgical considerations play a crucial role in ensuring the device's effectiveness and the patient's safety. These considerations not only encompass the procedural aspects but also focus on patient assessment and the strategy employed throughout the implantation. A well-planned surgical procedure can lead to better patient outcomes, reduced complications, and a smoother recovery process, making it an essential component of cardiac care related to S-ICDs.
Preoperative Assessment
Before heading into the operating room, a thorough preoperative assessment of the patient is necessary. This stage primarily involves understanding the individual patient's health status and their suitability for an S-ICD.
- Medical History: The doctor will review the patient’s complete medical history, including previous cardiac events, comorbidities, and any medication that could influence the surgical procedure or device functioning.
- Physical Examination: A comprehensive physical examination helps assess the patient’s overall cardiac and physical health, ensuring that the surgical team can gauge potential risks during the surgery.
- Imaging and Diagnostic Tests: Doctors may require imaging studies, such as echocardiograms or CT scans, to visualize the thoracic anatomy and plan the surgery accordingly. Additionally, electrophysiological studies might be performed to understand arrhythmias better.
- Informed Consent: It's also vital for patients to fully understand the procedure, potential risks, and expected outcomes. Gathering informed consent reflects the principles of patient autonomy and shared decision-making.
This preoperative phase lays a solid ground for the surgical steps to follow. A careful evaluation can facilitate identification of any factors that could complicate the implant.
Surgical Procedure Steps
The implantation of an S-ICD involves a series of methodical steps, which must be executed with precision to ensure optimal device placement and function. The surgical process can typically be outlined as follows:
- Anesthesia Administration: Patients usually receive general anesthesia or conscious sedation, depending on the complexity of the case and the surgeon’s preference.
- Incision: A small incision is made beneath the left axilla, allowing for the introduction of the device and lead. The location is chosen to minimize visibility and aesthetic impact.
- Device Preparation: The S-ICD is prepared and tested for functionality before insertion. This step includes checking battery status and lead integrity.
- Lead Insertion: The lead is carefully inserted beneath the skin and subcutaneous tissue, allowing for adequate placement without entering the myocardium.
- Device Attachment: Once the lead is positioned properly, it is attached to the S-ICD device. Surgeons make sure it’s secure and functions as required.
- Suturing: The incision site is closed, usually with absorbable sutures or staples, ensuring that the device is well-protected under the skin.
- Post-Operation Monitoring: Patients are monitored closely in the recovery area following surgery to watch for any immediate complications.
Each step in the surgical procedure is vital. Attention to detail ensures that the S-ICD is not only placed correctly but operates effectively, which is key for the patient’s safety.
"In cardiac procedures like S-ICD implantation, thoroughness in surgical planning and execution can significantly influence long-term outcomes."
Overall, the surgical considerations surrounding S-ICD implantation provide a framework for clinicians to make informed decisions, enhancing patient care and upholding safety protocols in a potentially life-saving intervention.
Postoperative Management
Postoperative management is a critical phase in the care of patients who have received a subcutaneous implantable cardioverter defibrillator (S-ICD). The journey does not end once the device is successfully implanted; rather, it shifts focus to how the patient adjusts, heals, and integrates this technology into their daily lives. Ensuring that patients receive diligent follow-up care is fundamental in preventing complications and enhancing overall outcomes.
Monitoring and Follow-Up
Effective monitoring post-surgery involves a systematic approach to patient care. Initially, monitoring includes regular visits to the physician to evaluate the function of the S-ICD and ensure the site of implantation is healing without issues. The frequency of these visits may vary but often occurs within the first few weeks after surgery, and should continue over the long term.
Common parameters evaluated during follow-up visits might include:
- Device performance: Regular checks to ensure the defibrillator is operating correctly and that all settings are appropriate for the patient's needs.
- Electrode integrity: To assess if the electrodes implanted under the skin maintain their position and functionality.
- Patient symptoms: Monitoring for any arrhythmic events or complications like chest pain, shortness of breath, or unusual sensations helps provide a comprehensive understanding of the patient’s recovery.
- Device programming adjustments: There may be a need to fine-tune the device settings based on the patient’s evolving needs.
The importance of robust follow-up can't be overstated; it can detect any issues early, allowing for timely interventions.
Potential Complications and Resolution Strategies
While S-ICDs generally show promise, complications can arise, and being prepared to address them is key. Potential complications may include:
- Device infection: Surgical sites may become infected, necessitating prompt treatment, often with antibiotics.
- Displacement of the device: If the device shifts from its original position, this can impair its ability to function correctly. Surgical intervention may be required to reposition or replace the device.
- Device malfunction: Electronic failures or programming issues can occur, necessitating device reprogramming or replacement.
Strategies to manage these complications should encompass:
- Education for patients and caregivers: Informing them about signs of infection or malfunction boosts early detection.
- Prompt medical evaluation for symptoms: Patients should be encouraged to report any new or unusual symptoms immediately.
- Regular follow-ups: As previously mentioned, this ensures monitoring of the device and the patient's overall condition.
"Timely detection and intervention can significantly alter the trajectory of recovery and enhance life quality for those with S-ICDs."
In summary, postoperative management of S-ICD patients is not solely about monitoring but rather creating a supportive framework that emphasizes patient education, prompt intervention, and long-term follow-up. This holistic approach not only addresses potential complications but also reassures patients as they adapt to living with an S-ICD.


Future Directions
The landscape of cardiovascular care is continually evolving, and the subcutaneous implantable cardioverter defibrillator (S-ICD) is no exception. Future directions in this field underscore the significance of innovation and ongoing research, aiming to enhance patient outcomes and device capabilities. Understanding these developments is crucial for medical professionals, researchers, and patients alike, as they offer insights into how technology and clinical practices are about to change.
Innovations in Device Technology
As technology progresses, the S-ICD systems are anticipated to undergo remarkable transformations. The shift toward miniaturization of patient devices is one of the cornerstones of future innovations. This means future S-ICDs may become even smaller, allowing for easier implantation and less invasiveness, which can result in a more comfortable experience for patients.
Increased connectivity is also on the horizon with advancements in telemedicine. Imagine S-ICDs equipped with remote monitoring features. they could transmit vital metrics to healthcare providers in real time, allowing for precocious interventions when irregularities are detected.
Some possible innovations include:
- Advanced battery technologies: Longer-lasting batteries could significantly reduce the frequency of surgical replacements.
- Smart algorithms: Developing more refined algorithms that enhance arrhythmia detection and minimize false shocks would be a game-changer — essentially improving patient safety and experience.
- Biocompatible materials: The materials used in these devices are set to evolve as well, aiming to reduce complications related to tissue reactions and promote better integration with body systems.
These innovations could drastically improve the functionality, safety, and comfort of S-ICDs in the years to come.
Research and Development Trends
The research landscape surrounding S-ICDs is dynamic and continuously adapting. Key trends in this field spotlight increasing engagement within biomedical engineering initiatives and clinical trials that focus on patient outcomes and personalized medicine.
There is a growing body of literature exploring diverse patient demographics to understand differences in responses to S-ICDs. For example, how do age, sex, or comorbid conditions impact the efficacy and acceptance of these devices? Such insights are essential, as they allow physicians to tailor interventions according to individual patient profiles.
Ongoing studies are also examining:
- Long-term efficacy: Evaluating the durability and performance of S-ICDs over extended timeframes helps to establish a clearer picture of their effectiveness.
- Quality of life metrics: Assessing how living with an S-ICD affects daily activities, mental health, and overall well-being is pivotal in evaluating device success beyond clinical outcomes.
- Technological convergence: The collaboration between cardiac devices and mobile technology is another area garnering research attention, paving the way for integration with consumer health tech.
Patient Perspectives
Understanding patient perspectives in the context of subcutaneous implantable cardioverter defibrillators (S-ICDs) is pivotal. It brings to light how individuals feel before, during, and after the implantation of such devices. Insights from patients offer invaluable feedback regarding their experiences, fears, and expectations.
Living with an S-ICD
Living with a subcutaneous ICD can be a unique journey for patients. At first glance, the thought of having a device embedded under the skin may seem quite daunting. Many individuals express anxiety about the procedure and its aftermath. However, once they actually experience the benefits and gain familiarity with the device, many report a sense of relief.
One of the notable aspects that patients appreciate is the relative ease of life post-implantation. They often note that the device, which is less invasive than traditional ICDs, leads to reduced levels of discomfort and hassle. Patients have described it as feeling like an additional layer of skin rather than a clunky piece of technology. Adjusting to the presence of the ICD often requires time, but many find comfort in knowing it is there to protect them from sudden cardiac arrest.
"It’s like having a guardian angel. You forget it’s there, but it’s always watching over you," reflects a patient who had undergone the procedure.
Maintaining a routine, staying active, and engaging in social activities become essential aspects of life for these patients. Nevertheless, some patients have voiced concerns about restrictions, especially regarding physical activities. They may wonder what sports or movements are considered safe. Most healthcare professionals encourage adopting a proactive yet cautious attitude, promoting a blend of imagination and common sense in choices of activity.
Patient Education and Resources
Patient education is an ongoing process. After receiving an S-ICD, individuals often necessitate comprehensive guidance on managing their new normal. There are several resources available to assist patients in understanding the device and optimizing their health.
Key educational subjects typically include:
- Understanding the Device: Patients should have a fundamental grasp of how the S-ICD functions, including its monitoring capabilities and response mechanisms.
- Lifestyle Adjustments: Guidance on lifestyle changes post-implantation can be critical. Patients might need advice on activities to avoid or adapt, such as certain exercises or contact sports.
- Regular Follow-Up: Emphasizing the importance of routine check-ups ensures that the device is functioning properly and any issues can be addressed early on.
- Support Networks: Connecting with other patients who have undergone similar experiences can provide emotional support, making isolation feel less daunting. Online forums, community groups, and healthcare institutions often host support groups where individuals can share stories, advice, and encouragement.
By utilizing available resources and engaging in proactive learning, patients can significantly enhance their overall experience and quality of life with an S-ICD. Exploring platforms like reddit.com, where experiences are shared freely, or consulting further details on Wikipedia can open doors to enriching discussions that benefit both patients and caregivers.
Education translates to empowerment, allowing individuals to navigate the complexities of living with an S-ICD confidently.
Epilogue
In wrapping up our discussion about subcutaneous implantable cardioverter defibrillators, it's crucial to underscore the significant elements that have emerged throughout the exploration of this device. The subcutaneous ICDs, as a modern alternative to traditional transvenous ICDs, bring forth an array of benefits that extend far beyond mere technological advancement. As we’ve established in prior sections, they play a pivotal role in enhancing patient safety by reducing infection risks, as there's no intravascular component involved. This aspect fundamentally alters the post-operative experience for patients, allowing for a smoother transition back to daily life, accompanied by a peace of mind that cannot be overlooked.
Moreover, it’s evident that the design and operational mechanics of S-ICDs pave the way for a paradigm shift in arrhythmia management. The minimization of surgical complications not only underscores operational efficacy but also encourages broader adoption among healthcare providers. The intersection of innovation and patient comfort resonates throughout the entire process, from the initial assessment to ongoing management. Therefore, acknowledging these points lies at the heart of understanding the true impact of S-ICDs in cardiac care.
The conversations surrounding limitations are equally vital. Recognizing device displacement issues or the nuances of arrhythmia detection allows clinicians to tailor their approaches, ensuring that patients receive the most appropriate level of care according to individual circumstances. Such considerations, as discussed earlier, promote a balanced view about the role these devices occupy in modern medicine.
Summary of Key Points
- Improved Patient Safety: Reduced risk of infections due to the absence of intravascular components.
- Technological Advancements: Innovative design leads to enhanced operational mechanics.
- Surgical and Post-Operative Benefits: Lower complication rates and improved patient comfort.
- Consideration of Limitations: Acknowledging device issues allows for more tailored patient care.
- Future Implications: S-ICDs represent a shift in arrhythmia management that merits ongoing research and development.
Implications for Future Practice
The significance of subcutaneous ICDs in the evolving landscape of cardiac care cannot be understated. As healthcare professionals continue to integrate these devices into clinical practice, it is essential to stay sharp on the latest insights and developments surrounding S-ICDs. Continuous education will empower clinicians to understand the subtleties of device functionality, usage scenarios, and patient outcomes.
Additionally, ongoing research into innovations in device technology and trends in development will undoubtedly refine existing practices. Future training programs and clinical guidelines should encompass a wider breadth of information regarding potential complications, tailored monitoring strategies, and long-term patient management solutions.
The pathway forward is marked with promise, and a commitment to understanding the impacts of these devices on patient care will facilitate a smoother embrace of new techniques and guidelines as they arise.
As our understanding deepens, so too will the capacity to provide the best possible outcomes for patients entrusted to our care.







